The Comprehensive Guide to Benign Concentric Annular Macular Dystrophy (BCAMD)
Benign Concentric Annular Macular Dystrophy (BCAMD), colloquially known as Bull’s Eye Macular Dystrophy, is a rare but distinct phenotype in the spectrum of retinal disorders. While the term “benign” was historically assigned due to the preservation of central vision in early adulthood, modern longitudinal studies and clinical experience suggest a more complex, progressive nature that clinicians and patients must navigate together.

Pathophysiology and Presentation
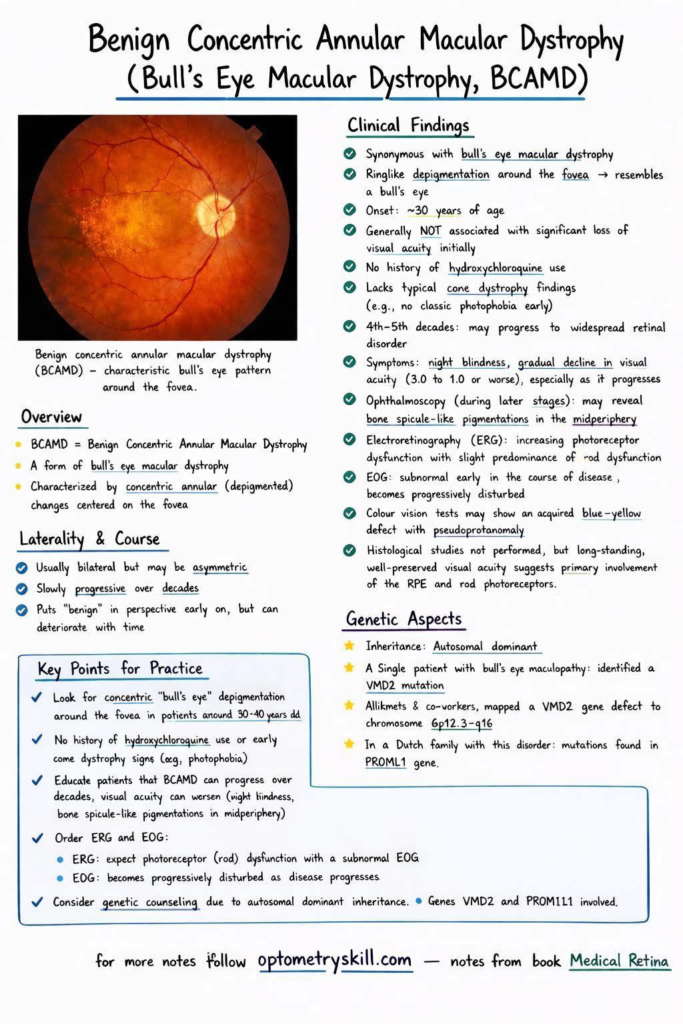
The hallmark of BCAMD is its striking ophthalmoscopic appearance: a ring-like, concentric zone of depigmentation surrounding the fovea. This “bull’s eye” pattern represents the atrophy of the Retinal Pigment Epithelium (RPE) and the overlying photoreceptors.
The Timeline of Progression
Unlike many aggressive childhood dystrophies, BCAMD typically manifests its first clinical signs around the age of 30.
- Early Stage: Visual acuity often remains high (frequently 20/20 or 20/25 initially). At this stage, the condition may be an incidental finding during a routine fundus exam.
- The Decadal Shift: As patients enter their 40s and 50s, the “benign” status often gives way to a more widespread retinal disorder. The atrophy may expand from the macula into the midperiphery.
- Late Symptoms: Patients eventually report a significant decline in visual acuity and the onset of night blindness (nyctalopia), signaling that the rod system is increasingly compromised.
Diagnostic Criteria & Differential Diagnosis
One of the most critical challenges in managing BCAMD is differentiating it from other conditions that cause bull’s eye maculopathy.
The “Plaquenil” Question
Perhaps the most vital clinical distinction is ruling out Hydroxychloroquine (Plaquenil) toxicity. Because the fundus appearance can be nearly identical, a thorough medication history is mandatory. BCAMD is diagnosed in the absence of such toxic exposure.
Electrophysiological Markers
To move beyond a visual diagnosis, clinicians rely on objective electrophysiological testing:
- Electrooculography (EOG): This test often shows subnormal results very early in the disease course, indicating early RPE dysfunction even when the patient feels their vision is “perfect.”
- Electroretinography (ERG): As the disease advances, ERG reveals increasing photoreceptor dysfunction.Interestingly, BCAMD often demonstrates a slight predominance of rod dysfunction over cone dysfunction, explaining the later onset of night blindness.
- Color Vision: Patients may show a specific acquired blue-yellow defect known as pseudoprotanomaly.
The Genetic Blueprint: From VMD2 to IMPG1
BCAMD follows an autosomal dominant inheritance pattern, meaning an affected individual has a 50% chance of passing the condition to each offspring.
Traditional clinical notes emphasize the VMD2 gene (on chromosome 6), but recent molecular breakthroughs have refined our understanding. Research increasingly points to the IMPG1 gene (Interphotoreceptor Matrix Proteoglycan 1) as a primary culprit.
- VMD2 (BEST1): Historically mapped to chromosome $6p12.3-q16$, this gene is critical for RPE health.
- IMPG1 (6q14.1): Recent studies (into 2026) have identified IMPG1 mutations as a major cause of Bull’s eye maculopathy. The IMPG1 gene encodes a protein that supports the “scaffold” between photoreceptors and the RPE.When this protein fails, the photoreceptors lose their structural support, leading to the signature annular atrophy.
- PROML1 Gene: Mutations here have also been linked to specific familial clusters, particularly in European cohorts.
Clinical Management & Patient Counseling
Because there is currently no “cure” for BCAMD, management is focused on monitoring, education, and supportive care.
1. Longitudinal Monitoring
Patients should undergo annual or bi-annual exams including fundus photography and Optical Coherence Tomography (OCT) to track the “ring” of depigmentation.
2. Managing Expectations
It is essential to explain that while they may see well now, BCAMD is a slowly progressive condition. The term “benign” should be contextualized; it refers to the slow speed of the disease compared to more rapid dystrophies, not its lack of long-term impact.
3. Genetic Counseling
Because of the 50% inheritance risk, families should be offered genetic counseling. Modern Next-Generation Sequencing (NGS) panels can now pinpoint specific IMPG1 or VMD2 mutations, which can help in screening family members before clinical symptoms appear.
Key Summary
- Look for: Concentric “bull’s eye” depigmentation in patients aged 30–40.
- Rule out: Hydroxychloroquine use and classic cone dystrophy.
- Order: Baseline and serial ERG/EOG to monitor the transition from macular to widespread retinal involvement.
- Update: Stay aware of IMPG1 as a central genetic target in modern diagnostic panels.
Final Note: While BCAMD allows for many years of stable vision, the transition to widespread retinal dysfunction in later life highlights the need for consistent ophthalmic follow-up and empathetic patient communication.
Disclaimer: This article is for educational purposes and based on clinical notes from Medical Retina. Always consult with a retinal specialist for personalized medical advice.
Fahmina is a qualified optometrist. She founded OptometrySkills.com to make professional-grade eye care knowledge accessible to practitioners and patients alike.
